 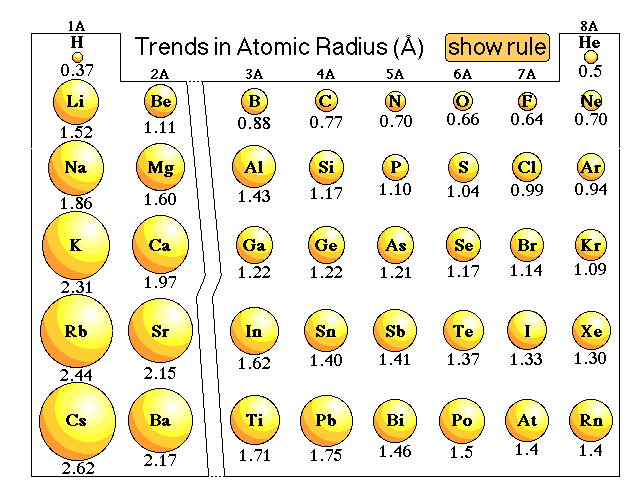
That means its electrons are more strongly attracted to its nucleus, hence the electronic cloud is more compressed, which results in a smaller size. Why does carbon have a larger atomic radius than oxygen?īoth are in the same period, but oxygen has a higher nuclear effective charge than carbon. Helium/Van der Waals radius What is the increasing atomic radius of carbon? 
As we can see, only Potassium has four electron shells, therefore Potassium has the largest atomic size. The potassium atom has an extra electron shell compared to the sodium atom, which means its valence electrons are further from the nucleus, giving potassium a larger atomic radius.ĭoes potassium have a larger atomic radius than oxygen?Īn atom’s size is defined by the distance from the nucleus to the valence (outer) shell of electrons. 
Does potassium have a larger atomic radius? Since C comes before O in the second period, O will have a smaller atomic radius than C. We know that atomic radius decreases from left to right within a period due to the increase in effective nuclear charge. Which has a greater atomic radius carbon or oxygen? Thus, helium is the smallest element, and francium is the largest. As can be seen in the figures below, the atomic radius increases from top to bottom in a group, and decreases from left to right across a period. Which element has the largest atomic radius list?Ītomic radii vary in a predictable way across the periodic table. 8 Which is the most abundant chemical element in the universe?.7 What is the atomic radius of boron and lithium?.6 What is the atomic radius of nitrogen?.4 What is the increasing atomic radius of carbon?.3 What is the atomic radius of potassium?.2 Does potassium have a larger atomic radius?. 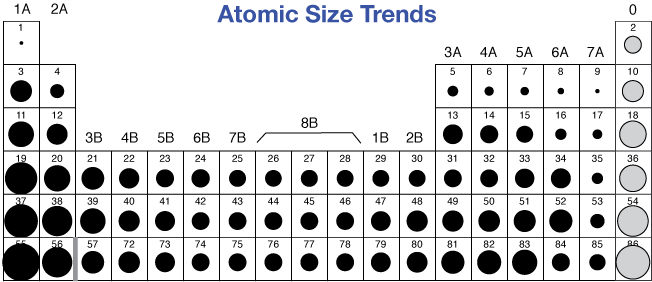
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
